The Comprehensive History of Soap During the Long 19th Century
The Evolution of Soap-Making Technology
The Leblanc Process and Its Industrial Impact
The Leblanc process, developed by French surgeon and chemist Nicolas Leblanc in 1791, transformed the production of lye (sodium hydroxide) and soda ash (sodium carbonate)—essential ingredients for soap-making and glass manufacturing. The process began with sodium chloride (common salt), which was converted into sodium sulfate by reacting it with sulfuric acid. The sodium sulfate was then heated with limestone (calcium carbonate) and coal to produce sodium carbonate. This innovation dramatically reduced costs, increasing soap availability during the Industrial Revolution and paving the way for the transition from artisanal to industrial-scale soap production. (Britannica) & (Chem Europe)
However, the Leblanc process came with significant environmental costs. It released hydrogen chloride gas, contributing to air pollution, and generated calcium sulfide waste, which caused foul odors and contaminated groundwater. These environmental hazards led to public health concerns and the implementation of governmental regulations in industrial regions. Ultimately, the Solvay process replaced the Leblanc method, not only because it was more economically efficient but also due to growing awareness of industrial pollution. This transition marked one of the earliest efforts to balance industrial progress with environmental responsibility.
The Solvay Process: Revolutionizing Soap-Making and Addressing Pollution
The Solvay process, developed in 1861 by Belgian chemist Ernest Solvay, revolutionized the production of soda ash (sodium carbonate), a vital ingredient in soap-making. This innovative method combined sodium chloride (brine) and calcium carbonate (limestone) with ammonia, which was recycled within the process, making it highly efficient and cost-effective. The Solvay process replaced the older Leblanc process, which, although effective, was costly and caused significant environmental damage, including hydrochloric acid emissions and calcium sulfide waste. (Solvay & Science Direct
While the Solvay process reduced costs and improved efficiency, it also brought its own environmental challenges, such as saline contamination of nearby water sources due to waste brine. In response, manufacturers adopted measures like ammonia recycling to mitigate some of these issues, making the process relatively more sustainable. However, the environmental impacts of soda ash production underscored the need for industrial pollution control—a concern that would become increasingly important in the 20th century.
The Innovations of Ludwig Mond
German-born British chemist and industrialist Ludwig Mond made significant contributions to refining the Solvay process, enhancing its efficiency and sustainability. Collaborating closely with Ernest Solvay, Mond developed advanced techniques for recycling ammonia within the process, which reduced production costs and minimized environmental waste. These improvements solidified the Solvay process as the dominant method for soda ash production in the late 19th century.
Image from Electrochemistry Europe
Mond’s influence extended beyond the soap industry, leaving a lasting impact across various sectors of industrial chemistry. His dedication to improving the sustainability of the Solvay process reflected a growing awareness of environmental issues in industrial operations—a legacy that continues to shape chemical engineering practices today.
Together, the contributions of Chevreul and Mond illustrate how the intersection of scientific research and industrial innovation drove advancements in both the quality and accessibility of soap, paving the way for modern soap-making.
Scientific Breakthroughs in Soap Chemistry
The Contributions of Michel Eugène Chevreul
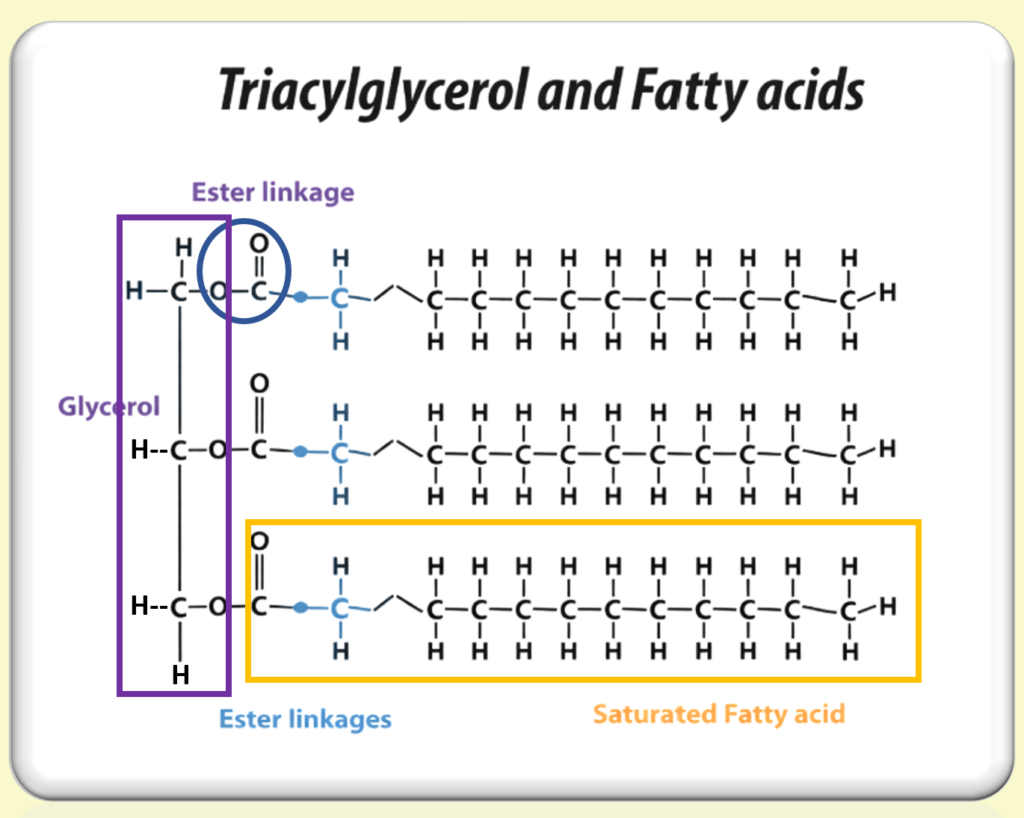
Michel Eugène Chevreul, a pioneering French chemist, made transformative contributions to soap-making in the 19th century through his groundbreaking research on fatty acids. In 1823, Chevreul published his seminal work on the chemistry of fats, revealing that fats are composed of glycerol (glycerin) and fatty acids. This discovery was instrumental in understanding the saponification process—the chemical reaction in which fats react with alkali to produce soap.
Chevreul’s research not only deepened the scientific comprehension of soap-making but also had far-reaching practical implications. By isolating specific fatty acids, such as stearic and oleic acids, he enabled soap-makers to refine their methods, leading to the production of soaps with superior cleansing properties, consistency, and lather. His work laid the foundation for modern soap chemistry, solidifying his legacy as one of the most influential figures in the history of the industry. (Lipid Library & Princeton University)
Socioeconomic and Regulatory Developments
Soap Taxation and the Guilds’ Role in Repeal Efforts
In the early 19th century, European soap production faced a formidable obstacle: steep government-imposed taxes. In Britain, soap had been taxed since the 17th century, initially to generate revenue for the monarchy. By the 19th century, these taxes had grown into a significant financial burden, turning soap into a luxury that many working-class families could scarcely afford. The tax applied not only to finished soap but also to its essential ingredients, further driving up costs and limiting production.
Guilds of skilled soap-makers became pivotal in the movement to repeal these taxes. They highlighted how the high cost of soap adversely impacted public health, especially in rapidly growing urban areas where hygiene and sanitation were critical for controlling disease. Reformers, industrialists, and public health advocates joined the guilds, building a compelling case for change. Their efforts bore fruit in 1853 when Britain repealed the soap tax, marking a turning point. This milestone made soap affordable and widely available, fueling a surge in production and contributing significantly to improved hygiene and public health standards.
Guilds and the Professionalization of Soap-Making
Soap-making guilds played a crucial role in regulating and advancing the craft throughout the Long 19th Century. These guilds established quality standards, protected trade secrets, and advocated for the interests of their members, including the repeal of burdensome taxes. In both Italy and France, guilds were instrumental in preserving traditional soap-making practices while encouraging innovation in production methods.
As industrialization progressed, many guilds evolved into professional associations, adapting to the changing landscape of the soap industry. Their efforts not only protected the heritage of the craft but also elevated soap-making to a respected profession, paving the way for the modern artisanal soap movements we see today.
Regional Soap-Making Traditions
The Rise of Italy and France as Soap-Making Centers
Italy’s renowned Noble Naples soap (Savon de Naples) was well-established long before France’s Savon de Marseille rose to prominence. The tradition of soap-making in Naples dates back to ancient Roman times, and by the 16th century, Naples had become a hub for producing some of the highest-quality soaps in Europe. These soaps, crafted from olive oil and lye, shared similarities with Marseille soap and were celebrated for their purity and mildness. Highly prized by European aristocrats, Naples soap became a luxury item across the continent.
Naples’ soap-making tradition flourished due to the region’s abundant olive oil production, which served as a key ingredient in their soap-making process. This tradition not only influenced neighboring regions but also played a crucial role in the development of soap-making in France. By the 17th and 18th centuries, Savon de Marseille had risen to prominence, in part due to the earlier success of Naples soap.
Marseille soap, like its Neapolitan counterpart, was made primarily with olive oil. However, it gained a reputation for being produced on a larger scale and for adhering to stricter production standards, set by the French king. This commitment to quality and consistency helped to standardize Marseille soap production, enabling it to reach a broader global market.
Thus, while Naples soap was the precursor and enjoyed significant popularity during its time, Marseille soap became the standard-bearer of European soap-making, eventually securing its place in history as one of the most famous soaps worldwide.
Other Advances and Trends in 19th Century Soap-Making
Commercial Branding
As soap production shifted to mass manufacturing, branded products emerged, with companies like Pears and Lever Brothers establishing recognizable names that emphasized both quality and accessibility.
Hygiene Awareness
As soap became more affordable, public health campaigns promoted its widespread use, linking cleanliness with disease prevention. This period saw soap firmly integrated into daily life.
Innovations in Fragrances and Formulations
Soap-makers began experimenting with a variety of fragrances and additives, leading to the creation of diverse products tailored to different consumer preferences.
Conclusion
The evolution of soap-making during the 19th century marked a pivotal transformation in both the craft and the industry. From the traditional methods upheld by guilds to the rise of industrial-scale production, this era laid the foundation for modern soap-making. The efforts of guilds to regulate the craft, alongside advancements in hygiene awareness, branding, and product innovation, helped establish soap as an essential part of everyday life. These changes not only improved accessibility and affordability but also contributed to the professionalization of soap-making, paving the way for the artisanal soap movements of today. As the industry continued to evolve, the legacy of the 19th century remains evident in the quality, variety, and cultural significance of soap around the world.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.