The Truth About Traditional Soap: Myths, Misconceptions, and Realities
Artisan Soap: The Real Deal
Introduction
Soap comes in many forms—soap, traditional soap, and what is commonly called conventional soap—but these terms do not all describe the same product. Traditional soap is true soap, while most modern “conventional” soaps are actually synthetic detergent cleansers, a difference that directly affects manufacturing methods and cost (click here to learn why).
The U.S. Food and Drug Administration acknowledges this distinction, noting that the majority of body cleansers sold today are detergent-based products (a cosmetic) rather than true soap. Despite widespread myths discouraging its use, traditional soap was once a household staple, used for everything from laundry to bathing and hair washing. Its decline in popularity began about a century ago, when detergents gradually replaced it.
True soap—sometimes labeled “natural”—traces back to a time when soap was made using rainwater-based lye solutions and animal fats, often as an annual or biannual farm chore. It’s understandable that older generations may associate traditional soap with harsh results from these early methods. However, the FDA does not define the term natural, nor does it regulate true soap as a cosmetic product.
My Initial Misgivings About True Soap
Years ago, while browsing a specialty shop selling handcrafted soap, I balked at the price and muttered to my husband, “I’m not paying THAT much for a bar of soap!” At the time, I had never used traditional soap. I had switched to shampoo at age fourteen because conventional bar soap felt harsh on my skin.
I mistakenly assumed there was no real difference between department-store soap and artisan soap beyond appearance. I viewed handcrafted soaps as overpriced luxuries rather than functional products. I couldn’t have been more wrong. Now that I craft artisan soap myself, I fully understand the reasons behind its cost. If you’ve ever wondered why artisan soap is priced differently, click here to learn more.
Ironically, it wasn’t until my husband raved about the first handmade soap I made that I finally tried it myself. I initially began making soap to control fragrance levels, as I’m hypersensitive to scents. My husband disliked unscented soap because it didn’t lather well. After retiring, I began making soap regularly—and now I never travel without it.
Why Artisan Soap Is the Real Deal
Artisan soap stands apart because it is genuine soap, not detergent marketed as soap. While detergent-based cleansers are not classified as soap by the FDA, they are still permitted to use the word soap on their labels, which understandably causes confusion.
The distinction lies in the ingredients. Products made with synthetic detergents are not true soap; they are cosmetic cleansers. Historically, detergent-based products could not legally be labeled as soap—yet today, many are.

Advertising Strategies and Misconceptions
Commercial manufacturers often promote detergent-based cleansers as superior to “lye soap,” capitalizing on fears of harshness. This misconception stems from early soap-making practices, which were imprecise and could result in caustic soap.
Modern soap-making is very different. When properly formulated, soap contains no excess lye. The FDA confirms that when soap is made correctly, no lye remains in the finished product.
Cosmetic regulations require manufacturers to list ingredients using INCI names, such as sodium tallowate or sodium cocoate. These terms describe saponified fats or oils, but they conveniently obscure the role of lye in production. This avoidance perpetuates fear-based misconceptions, causing people to associate “lye soap” with skin irritation or damage.
Marketers often exploit the word lye to suggest harshness, even though this perception is rooted in outdated soap-making methods. While earlier generations may recall homemade soap being rough, modern techniques ensure well-balanced, gentle soap.
Understanding how fats and fatty acids behave in soap was a turning point in the history of soapmaking. The work of Michel Chevreul helped soapmakers move beyond harsh, lye-dominant formulas toward more balanced and predictable soaps. To explore the scientific discoveries behind this shift, click here.
The Evolution of Soap Making
Soap making has evolved dramatically. Today’s artisan soaps are crafted with precision, using carefully calculated recipes that leave no lye in the final product—a fact acknowledged by the FDA. These soaps bear little resemblance to the unpredictable, caustic batches of the past and instead offer a gentle, effective cleansing experience.
The Tarnished Reputation of Traditional Soap
Despite these advancements, marketing narratives have unfairly tarnished traditional soap’s reputation. Myths and misinformation are often used to elevate detergent-based cleansers by comparison.
Curious to uncover the truth? Below, we’ll explore some of the most common myths surrounding traditional soap and explain why it still deserves a place in modern cleansing routines.
.
Shedding Light on Soap Misconceptions
My goal is education—not fear. After years of researching soap ingredients and formulations, I aim to help consumers understand the real differences between true soap, combo bars, and syndet cleansers, so they can make informed decisions—whether they choose my products or not. Education empowers choice.
The Role of INCI Names in Marketing
FDA regulations require cosmetic cleansers to list ingredients using INCI terminology. This allows companies to obscure the fact that sodium hydroxide or potassium hydroxide (lye) was used during production. Names such as sodium tallowate or sodium cocoate simply indicate that fats or oils were saponified using lye.
Ironically, while marketers criticize traditional soap for containing lye, they often avoid disclosing that combo bars—cleansers combining true soap with synthetic detergents—also rely on lye during manufacturing. One reason detergents are added to combo bars is to reduce soap scum in hard water, as detergents do not form soap scum the way true soap does.
Soap vs. Detergents: A Trade-Off
Both soap and detergent-based cleansers have trade-offs. True soap can form soap scum in hard water but is biodegradable and environmentally friendly. Detergent-based cleansers reduce soap scum but may raise environmental concerns, depending on the surfactants used.
Understanding these differences allows consumers to choose the product that best aligns with their needs and values.
It’s also worth noting that soap is fully biodegradable only when biodegradable fragrances and colorants are used.
Navigation
This page includes both accordion sections for brief, science-based explanations and clickable tiles that lead to more in-depth, dedicated pages. The accordions below address nine common myths surrounding traditional soap; expanding the accordion within each tile provides a concise overview, while clicking on the tile title opens a deeper exploration of that specific myth, including historical context and cited research. This layout allows readers to quickly understand the essentials and then dive deeper into the science behind any topic they choose.
Misconception: Traditional Soap is Harsh
A common myth about traditional soap is that it is harsh because it contains lye. While lye is an essential ingredient in soap making, it is not present in the finished product when the recipe is properly formulated. During the chemical process of saponification, lye reacts completely with fats or oils to form soap and glycerin, leaving no free lye behind to irritate the skin.
Even the FDA confirms: “When made properly, no lye remains in the finished product.”
This misconception largely stems from historical experiences with early “lye soap,” which could indeed be harsh due to the lack of precision in its preparation.
The Challenges of Early Soap Making
In pioneer-era soap making, lye was produced by leaching alkaline compounds from hardwood ashes. The strength of this homemade lye varied widely from batch to batch. Without accurate measuring tools or chemical knowledge, soap makers relied on crude testing methods—such as floating an egg or dissolving a feather—to estimate lye strength.
While these techniques allowed soap to be made, they often resulted in excess lye, producing soap that was overly caustic and irritating to the skin. Soap making was also labor-intensive and typically done only once or twice a year, further increasing the likelihood of inconsistent results. These early practices gave “lye soap” its long-lasting reputation for being rough and harsh.
Advances in Modern Soap Making
Significant advances in soap chemistry began in the late 19th century with the development of saponification (SAP) values, which identify the exact amount of lye required to fully react with specific fats and oils. This allowed soap makers to precisely balance their recipes.
Today, modern soap making benefits from accurate scales, reliable SAP data, and formulation tools that ensure consistency and safety. As a result:
No excess lye remains in properly made soap
The finished soap is non-caustic and skin-safe
Results are consistent and predictable
Modern traditional soaps are vastly different from early pioneer soaps
Conclusion
The belief that traditional soap is harsh because it contains lye is outdated and does not reflect modern soap making. When properly formulated, traditional soap contains no free lye and is neither caustic nor harsh. Unlike the unpredictable batches of the past, today’s artisan and traditionally made soaps are carefully balanced, consistent, and gentle—offering effective cleansing without the harshness once associated with early “lye soap.”
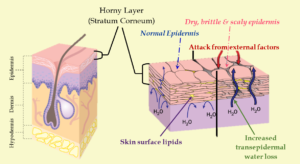
Misconception: Traditional Soap Strips Moisture from the Skin
A common belief is that traditional soap is inherently drying and removes too much moisture from the skin. This idea is largely rooted in outdated assumptions and experiences with harsh cleansing products, not with properly made soap itself.
When formulated correctly, traditional soap does not permanently strip the skin of moisture or damage its natural barrier. As a rinse-off product, soap cleanses the skin and is then removed, allowing the skin’s natural processes to resume.
Understanding Transepidermal Water Loss (TEWL)
Transepidermal Water Loss (TEWL) refers to the normal movement of water from within the body through the outermost layer of the skin. When the skin’s barrier is compromised—due to damage, over-cleansing, or harsh surfactants—TEWL can increase, leading to dryness, cracking, and discomfort.
I experienced this firsthand while dealing with severely dry, fissured skin. For years, my skin barrier struggled to recover, and while creams and ointments helped reduce moisture loss temporarily, they did not address the underlying imbalance.
What ultimately made a difference was switching to traditional, well-formulated soap and eliminating frequent use of detergent-based cleansers that left my skin feeling tight and stripped.
How Properly Made Soap Interacts with the Skin
Unlike many synthetic detergent cleansers designed to aggressively remove oils, traditional soap works differently. When properly made and used as intended:
Soap cleanses without leaving residual surfactants on the skin
It does not continue interacting with the skin after rinsing
It allows the skin’s natural lipid barrier to rebalance itself
Because soap is a rinse-off product, any temporary changes to the skin’s surface are short-lived. Healthy skin is a living, self-regulating organ and is capable of restoring its barrier function after cleansing.
The Bottom Line: Soap Is Not Inherently Drying
Traditional soap is not inherently drying, nor does it permanently disrupt the skin’s moisture barrier when properly formulated and used. The long-standing belief that soap “dries out the skin” is more accurately associated with over-cleansing, harsh detergents, or compromised skin barriers, rather than with true soap itself.
When balanced formulations are used and the skin is allowed to function naturally, traditional soap can cleanse effectively without working against the skin’s ability to maintain moisture.
Misconception: Traditional Soap Permanently Disrupts Skin pH
A common myth is that traditional soap damages the skin by disrupting its acidic mantle and weakening its natural defenses. While this claim is often repeated in marketing, it is not supported by long-term scientific evidence.
It is true that soap temporarily raises the skin’s surface pH during cleansing. However, this effect is short-lived and does not result in permanent damage to the skin’s protective barrier.
The Real Impact of Soap on Skin pH
Healthy skin typically has a slightly acidic surface pH, often around 4.5–5.5. When soap is used, the skin’s pH rises temporarily due to the alkaline nature of soap and water. Once rinsed away, the skin begins restoring its natural acidity.
Research shows that this recovery happens within hours, not days, and without lasting impairment. In a long-term study by Takagi et al. (2014), individuals who used soap-based cleansers and those who used mildly acidic cleansers for more than five years showed nearly identical skin pH values, demonstrating that soap does not impair the skin’s ability to regulate its pH over time.
What Actually Influences Skin pH
Skin pH is influenced by many factors, including:
Age
Environment and climate
Genetics
Overall skin health
Frequency and method of cleansing
While cleansing temporarily alters surface pH, the skin has local buffering mechanisms within the epidermis that quickly restore its natural balance. These processes are part of the skin itself and do not rely on internal organs or systems.
Skin’s Natural Ability to Recover
Skin is a living, resilient organ designed to adapt to everyday exposures—including water, sweat, and cleansing. Temporary pH shifts caused by washing are a normal part of daily life and are not harmful to healthy skin.
Much like rinsing with plain water, soap alters surface conditions briefly. Once cleansing ends, the skin resumes its natural regulatory functions and restores its protective barrier.
Debunking the Myth
The idea that traditional soap permanently damages the skin’s acid mantle is not supported by scientific evidence. When properly formulated and used as intended, traditional soap does not disrupt the skin’s long-term pH balance.
Claims that soap irreversibly harms the skin often confuse temporary surface changes with permanent damage. In reality, healthy skin is fully capable of rebalancing itself after cleansing—whether soap is used or not.
Misconception: Soap Permanently Disrupts the Skin Microbiome
A common misconception is that traditional soap harms the skin’s natural flora, also known as the skin microbiome. While cleansing does temporarily reduce surface microorganisms, this does not equate to permanent damage or imbalance when regular soap is used appropriately.
The Science Behind Soap and the Skin Microbiome
The skin’s microbiome is made up of a diverse population of microorganisms that live on and within the outer layers of the skin. This ecosystem is dynamic and resilient, constantly changing in response to daily activities such as washing, sweating, and environmental exposure.
When soap is used, the number of surface microbes is temporarily reduced, along with oils and debris. Research shows that this reduction is short-lived, and the skin’s microbial population re-establishes itself naturally over time.
Misconception: Sharing Bar Soap Spreads Germs
Many people feel uncomfortable using bar soap—especially shared bar soap—because of concerns about germs and disease transmission. This “ick factor” is understandable, but it is based more on perception than on scientific evidence.
What the Science Says
For decades, researchers have studied whether bacteria present on a used bar of soap can transfer to another person during handwashing. In a well-known study by Bannan and Judge (1965), bars of soap were deliberately contaminated with high levels of bacteria. Even under these exaggerated conditions, no bacteria were transferred to users’ hands during washing.
The reason lies in how soap works. Soap does not act by sterilizing the skin. Instead, it loosens and lifts microbes, allowing them to be carried away with water during rinsing. The brief contact time and continuous rinsing prevent bacteria from remaining on the skin or being transferred from the soap itself.
Additional Research Confirms the Findings
Later studies reinforced these conclusions. Research by Heinze and Yackovich (1988) again demonstrated that bacteria placed on bar soap did not transfer to hands during normal use—even when contamination levels were far higher than would occur in everyday settings.
Together, these studies show that bar soap does not serve as a vehicle for disease transmission under normal conditions of use.
Why Bar Soap Doesn’t Spread Germs
Soap is used with running water, which rinses microbes away
Contact between soap and skin is brief
Bacteria do not readily survive or transfer during the washing process
Soap works through physical removal, not by leaving residue behind
Conclusion
Scientific evidence does not support the idea that bar soap spreads germs or transmits disease. Multiple studies have shown that bacteria placed on bar soap are not transferred during handwashing.
The belief that bar soap is unhygienic is rooted in discomfort, not data. When used properly with water and rinsing, bar soap is a safe and effective way to cleanse the skin.
Misconception: Antibacterial Soap Cleans Better Than Regular Soap
A common belief is that antibacterial soap is more effective than traditional soap at preventing illness. However, scientific evidence and public health guidance do not support this claim for everyday use.
Not all illnesses are caused by bacteria—many are caused by viruses, against which antibacterial ingredients offer no added benefit. For routine handwashing, regular soap and water are just as effective as antibacterial soap.
Why Soap and Water Are Highly Effective
Soap works by physically removing germs, not by killing them. The soap molecules loosen dirt, oils, bacteria, and viruses from the skin so they can be rinsed away with water.
Health organizations consistently emphasize this mechanical action:
The CDC states that washing hands with soap and water for at least 20 seconds is one of the most effective ways to prevent the spread of illness.
The Mayo Clinic confirms that over-the-counter antibacterial soaps are no more effective than regular soap and water for everyday handwashing.
The Cleveland Clinic similarly recommends regular soap for routine hygiene.
What the FDA Says About Antibacterial Soaps
The FDA has reviewed antibacterial soaps extensively and found no evidence that they provide additional health benefits over plain soap and water for consumer use.
As a result, certain antibacterial ingredients—such as triclosan—were restricted or removed from many over-the-counter products due to concerns about:
Lack of added effectiveness
Potential contribution to antibiotic resistance
Long-term safety
These actions were not taken against regular soap, which remains a recommended and effective cleansing option.
Clarifying an Important Distinction
Regular soap: Cleans by lifting and rinsing away germs
Antibacterial soap: Regular soap with added antimicrobial agents
Syndet cleansers: Synthetic detergent bars or liquids, which may or may not contain antibacterial ingredients
Antibacterial performance depends on added agents, not on whether a cleanser is soap or syndet—and those added agents have not been shown to improve everyday hygiene outcomes.
Conclusion
Traditional soap is not less effective than antibacterial soap for routine cleansing. In everyday situations, plain soap and water are fully sufficient and are consistently recommended by major health organizations.
The belief that antibacterial soap provides superior protection is a misconception. What matters most is proper handwashing technique, adequate contact time, and thorough rinsing—not the presence of antibacterial additives.
Misconception: Tallow and Lard Are “Low-Quality” Soap Ingredients
Traditional soaps made with animal fats such as tallow or lard are sometimes dismissed as outdated or inferior. In modern marketing, plant oils are often portrayed as automatically superior, while animal fats are framed as something to avoid.
Historically and chemically, however, this perception is not accurate.
Animal Fats and Plant Oils in Soap Making
Both animal fats and plant oils are composed primarily of triglycerides, which react with lye during saponification to form soap molecules and glycerin. Once this reaction is complete, the resulting soap functions the same way regardless of whether the original fat came from an animal or a plant.
In properly formulated soap, no lye remains, and the cleansing behavior is determined by the fatty acid profile of the oils used—not their source.
Many artisan soaps, including mine, are made with a small intentional excess of oils (often called a superfat). This means a very small portion of the original oils remains unsaponified. This does not make the soap greasy, but it can reduce the tight or overly stripped feeling some people experience after washing.
Why Tallow Has Been Used for Centuries
Tallow, traditionally rendered from beef fat, has been used in soap making for centuries because it produces a hard, long-lasting bar with a stable, creamy lather. Its fatty acid composition—rich in stearic and palmitic acids—contributes to the structure and mildness of the soap itself.
While raw tallow contains components such as fatty acids, cholesterol, and other lipids that are also found in human skin, it is important to clarify that soap is a rinse-off product. These constituents do not remain on the skin long enough to function as active skincare ingredients in the way they would in leave-on products.
The Case for Lard
Lard, rendered from pig fat, has a fatty acid profile that produces a gentle, conditioning-feeling soap with excellent lather characteristics. Like tallow, it has been used historically because it makes a well-balanced bar that cleans effectively without excessive harshness.
Its value in soap lies in how it contributes to bar performance and user experience, not because it delivers nutrients to the skin.
Important Context: Soap vs Leave-On Products
Although tallow and lard contain skin-related lipids in their raw form, these components are not delivered to the skin in a meaningful way through soap. Soap is designed to cleanse and be rinsed away.
Any benefits from these fats in soap are functional, not therapeutic:
Bar hardness
Creaminess of lather
Mild cleansing feel
Leave-on products such as lotions, balms, or creams are where ingredients like fatty acids, cholesterol, and ceramides can play an active role on the skin.
Conclusion
Soaps made with animal fats such as tallow or lard are not inferior products. They are time-tested materials that produce stable, effective, and gentle cleansing bars when properly formulated.
Dismissing these ingredients as outdated overlooks both their historical role and their functional value in soap making. While soap is not a delivery system for skin nutrients, well-formulated traditional soap—whether made with animal fats or plant oils—remains a reliable and effective way to cleanse the skin.
Misconception: “No-Lye Soap” Is Possible
Claims about making soap without lye are common—but they are chemically incorrect. True soap cannot be made without lye. Soap is created through a chemical reaction called saponification, which requires an alkali. For bar soap, that alkali is sodium hydroxide; for liquid soap, it is potassium hydroxide.
Without an alkali, saponification cannot occur. No alkali means no soap—it really is that simple.
Why “No-Lye Soap” Is Chemically Impossible
Some products and methods are marketed as “lye-free,” most commonly melt-and-pour (M&P) soap bases. Melt-and-pour soap is pre-made, fully saponified soap that can be melted and reshaped. While the person using it does not handle lye themselves, lye was absolutely used to make the original soap base.
Because the lye has already reacted during manufacturing, it is no longer present as free lye in the finished base—but that does not make the soap “lye-free.”
The same misunderstanding applies when a bar of soap is dissolved in hot water and called “lye-free liquid soap.” The soap itself was still made using lye, and dissolving it does not change its chemical origin.
Marketing these methods as “no-lye soap” confuses process with chemistry.
Conclusion
There is no such thing as true soap made without lye. While some soap-making methods allow people to avoid handling lye directly, lye is always required somewhere in the process for saponification to occur.
So the statement holds true:
No lye, no soap.
Understanding this distinction helps separate accurate chemistry from marketing language—and clears up one of the most persistent myths surrounding traditional soap.
Misconception: Handmade Soap Is Always Made Only from Natural Ingredients
A common assumption is that handmade or traditional soap is automatically “all-natural.” In reality, the term natural has no formal definition for soap under U.S. regulations. The U.S. Food and Drug Administration does not define or regulate the terms natural or organic for soap, which means these words are often used as marketing descriptors rather than scientific classifications.
From a regulatory standpoint, soap may contain both naturally derived and synthetic ingredients, provided they are safe for use. This includes fragrances, colorants, and other additives commonly used in modern soap making.
.
Natural vs. Synthetic Ingredients in Handmade Soap
Some artisan soap makers choose to work exclusively with plant-derived oils, essential oils, herbs, and food-based colorants. These soaps are often described as natural soap. Others incorporate fragrance oils or FDA-approved synthetic colorants for reasons of stability, consistency, and design flexibility.
From a regulatory perspective, both approaches result in soap. The difference lies in ingredient choices, not in whether the product is considered soap.
Cost and Stability Considerations
Natural ingredients such as essential oils are typically more expensive and can vary in scent and performance due to natural fluctuations in plant chemistry. Natural colorants—such as herbs, clays, or food powders—often produce softer tones and may fade or change color over time.
For this reason, many soap makers choose FDA-approved synthetic colorants, which offer:
Color stability
Consistent results
Vibrant, long-lasting designs
Using these ingredients is a practical choice, not a shortcut.
“Synthetic” Does Not Mean “Unnatural in Origin”
Many synthetic materials still trace back to natural raw sources, even though they are processed or refined by humans. For example:
Glass is made from sand
Polyester is derived from petroleum
Coal originates from ancient plant matter
In soap making, synthetic colorants and fragrance components are often nature-identical, meaning they are chemically the same as compounds found in nature but produced in a controlled, purified form.
Colorants in Soap
Soap makers may choose from a range of approved colorants, including:
Micas
Titanium dioxide
Iron oxides
Ultramarines (not approved for lip products)
Natural colorants tend to produce muted, earthy tones, while synthetic colorants allow for brighter, more predictable results. Both options are widely used and considered safe when properly formulated.
Traditional Soap's Bum Rap
How do myths about anything get started? Generally, they’re rooted in partial truths. A part-truth is called a lie; so, myths are just that: untruths–not factual. Like all good myths, they get perpetuated and take on a life of their own, living in infamy. All of the following myths do make sense to me, in how they were started.
Many combination bars are made using lye during the manufacturing process. However, under FDA labeling regulations, ingredient lists reflect the final, finished ingredients — not the raw materials used to create them. As a result, you won’t see the word “lye” listed on properly made soap. I’m not required but I list it on my soap.
If you’ve never tried “real” (traditional) soap then you have nothing to compare your current detergent -based cleaner. If you have tried traditional soap, no need to even read this info; you already have discovered the world of “real” soap.
Guess what?
Multiple colonies of various bacteria live on the surface of the skin and even down into the hair follicles–ewww, right?
Are the good bacteria being wiped out by washing with alkaline soap??
This is something to think about if only in a public setting.
Studies have been conducted which answer this conundrum.
If you’ve heard that true soap is subpar to commercial cleansers, what are the reasons given? Feel free to share what you’ve heard or read in the REPLY section of this topic.

Even if FOs and synthetic colors are used, the preponderance of ingredients (the bulk) will be things sourced from nature.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.