Amphiphilic??
Heavens to Murgatroyd what kind of 😨 monster 😨 is an amphiphilic ? ? ?
Introduction:
An amphiphile (from the Greek amphis, meaning both, and philia, meaning love) is a chemical compound that possesses two opposing traits at the same time:
a hydrophilic (water-loving, polar) part
a lipophilic (fat-loving, non-polar) part
Because of this dual nature, these compounds are described as amphiphilic (also called amphipathic). Soap is one of the most familiar amphiphilic substances we encounter in everyday life.
Wikipedia link
Water is Sticky
. . .
And Not Very Good at Cleaning on Its Own
Water molecules are attracted to each other through hydrogen bonding, a property known as cohesion. Each water molecule has two slightly positive hydrogen atoms and one slightly negative oxygen atom, causing neighboring molecules to cling tightly together.
This cohesive force is strong enough to allow water droplets to pile up on a flat surface—such as a penny—forming a dome shape. Only when gravity overcomes this attraction does the water spill over.
Inside a body of water, molecules are pulled equally in all directions. At the surface, however, molecules experience an inward pull because there are fewer neighboring molecules above them. This creates surface tension, forming a flexible, skin-like barrier at the water’s surface.
While this property is fascinating, it also makes water inefficient at cleaning, especially oils and grease.
May the Force Be With You, Lucas . . . I Mean Surface Molecules
Surface tension can be strong enough for insects like water striders—or even spiders—to walk across water. Their weight is distributed in a way that doesn’t break the cohesive bonds.
A paperclip can float for the same reason—until surface tension is disrupted.
When it comes to cleaning, however, surface tension works against us. Cold water has stronger cohesion, making it harder for water to penetrate fabrics or break up oily grime. Hot water weakens these bonds, lowering surface tension and improving cleaning ability.
RED ROVER, RED ROVER, WE CALL SURFACTANT OVER
What's a Surfactant?
The term “surfactant” is a combination of three words: “surface-active-agent“(surfactant).
A surfactant is a substance that reduces surface tension between two materials—such as water and oil—allowing them to interact.
All soap is a surfactant, but not all surfactants are soap.
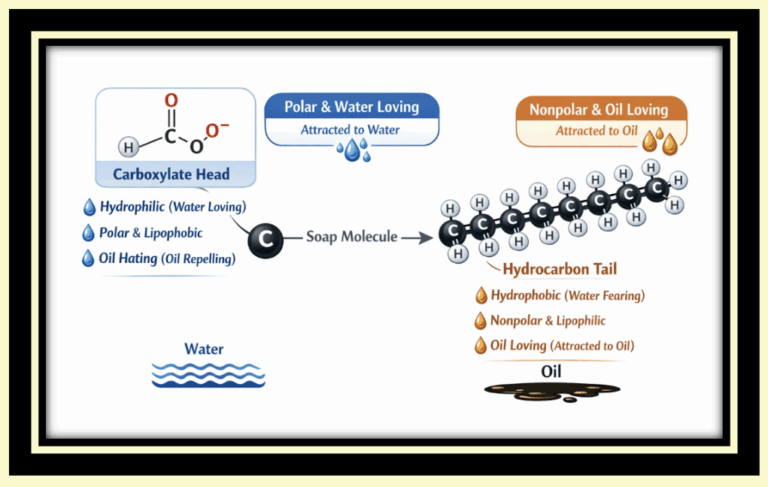
Traditional soap molecules have:
a hydrophilic head that interacts with water
a hydrophobic tail that avoids water and binds to oils
Photo from Wikipedia
This dual structure allows soap to bridge the gap between substances that normally don’t mix.
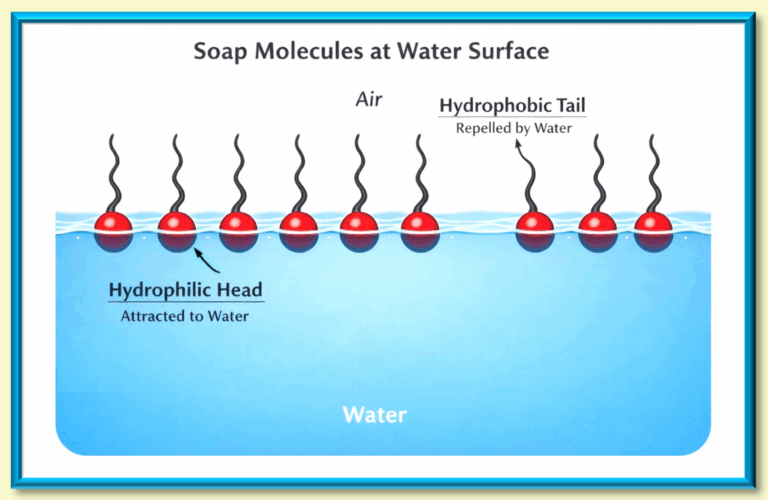
Think back to the game Red Rover. The chain of hands represents hydrogen bonds holding water molecules together. The child called over is the surfactant—breaking through the chain and disrupting surface tension.
Soap, Making Water Wetter . . .
(Yes, it’s already wet — but stick with me)l
The hydrophobic tail disrupts hydrogen bonding between water molecules, reducing surface tension. This allows water to spread more easily and reach grime trapped on skin, fabric, or dishes.
If you add soap to water where a spider is walking, surface tension collapses and the spider sinks. Not a great use of soap—but a dramatic demonstration of how it works.
Without soap, water alone struggles to remove oily dirt. Soap enables water to do what it can’t do by itself.
Soap: The Mediator Between Water and Oil
Making Them Play Nicely Together, Only to Send Them Down the Drain as Partners in “Grime.”
Oil is hydrophobic—it avoids water (like we are fearful of spiders and avoid them).
Since water and oil don’t mix, a mediator is needed to remove oily sweat and grime.
In the words of Snagglepuss: “Heavens to Murgatroyd! Exit, stage left!” Enter, stage right… soap!
Exit, stage left: Bye, bye oily grime and germs… and a bit of our skin’s natural oils.
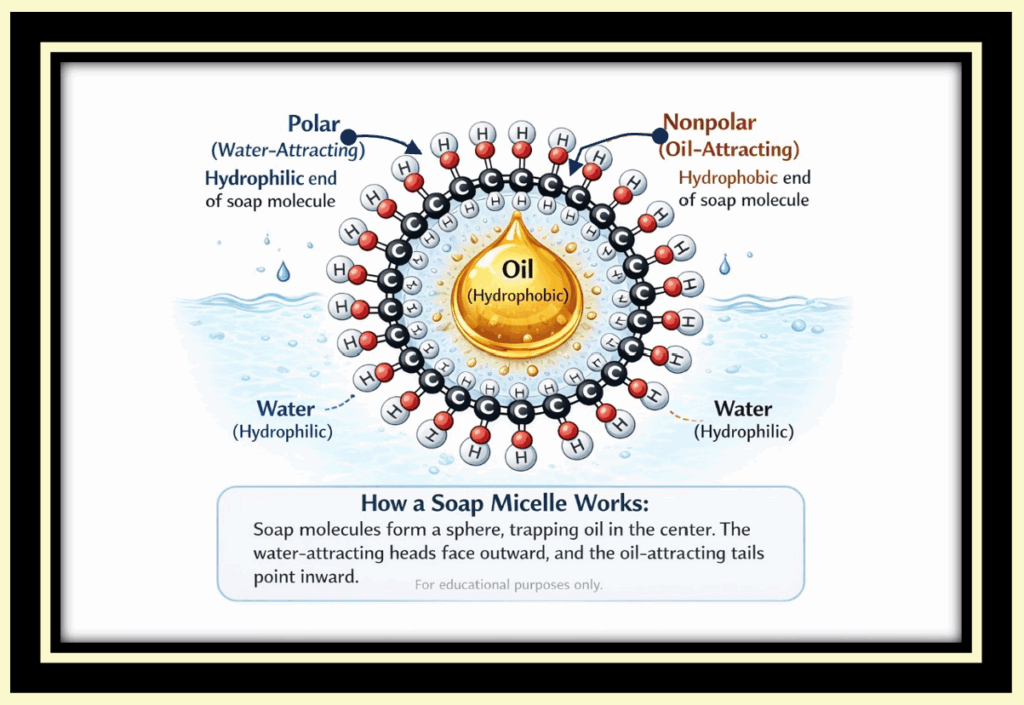
Soap doesn’t dissolve dirt; it surrounds it.
The hydrophobic tails embed themselves in oil and grease, while the hydrophilic heads face outward toward water. This forms a structure called a micelle, which traps the grime inside and allows it to be rinsed away.
Soap helps water and oil “play nicely together” long enough to be washed down the drain—along with some of the skin’s natural oils.
The Science behind Soap’s Cleaning Power: Amphiphilic by Nature
Soap is amphiphilic by design. Its ability to interact with both water and oil is what makes cleansing possible.
When soap is used:
hydrophobic tails bind to grease
hydrophilic heads bind to water
micelles form
grime is lifted and rinsed away
In short, soap lowers surface tension and physically removes dirt, making it an effective cleanser.
If the term micelle sounds familiar, you may recognize it from products marketed as micellar water—which rely on the same basic chemistry.
Soap micelle
If seeing the word “MICELLE” seems like déjà vu, you’re probably recalling commercials advertising “Micellar” water for cleansing the face.
Heinrich Gottlob Bertsch and the Birth of Synthetic Detergents
Chemist Heinrich Gottlob Bertsch (Wikipedia)
Heinrich Gottlob Bertsch (1897–1981) was a German chemist who developed the world’s first fully synthetic detergent. His work addressed soap’s limitations—especially poor performance in hard and salt water—by creating detergents that remained effective under a wider range of conditions.
In 1932, Bertsch developed Fewa, the first fully synthetic and mild detergent. Its success reshaped cleaning chemistry and laid the foundation for the modern detergent industry.
These innovations proved essential for industrial, military, and maritime use and eventually influenced household cleaning products worldwide.
From Traditional Soap to Synthetic Detergents — and Back Again
Synthetic detergents were first developed in Germany in the early 20th century. Detergent-based cleansers for body and hair later appeared in the United States and became widespread after World War II.
Over time, detergents dominated the market as manufacturing priorities and consumer habits shifted. In recent years, however, traditional soap has seen renewed interest, as people seek simpler formulations and a better understanding of how cleansing products actually work.
To explore how soap and detergents differ chemically—and why both still exist today—click below:
Cleaning Chemistry: Natural Soap vs. Lab-Made Detergents
From Soap to Detergents — and Back Again
Synthetic detergents were first developed in Germany (info above) in the early 20th century, driven by advances in industrial chemistry and the need for alternatives to fat-based soap. Detergent-based cleansers for body and hair later began appearing in the United States in the early-to-mid 20th century, roughly 80–100 years ago, becoming widespread after World War II. Over time, these products came to dominate the market as manufacturing priorities and consumer habits changed. In recent years, however, traditional soap has been experiencing renewed interest.
To learn more about why cleansing products shifted from soap to detergent-based formulations — and why traditional soap is now seeing a resurgence — click the link below to explore this topic in greater depth.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.