Cleansing Chemistry: Natural Soap vs. Lab-Made Detergents
How Genuine Soap Differs from Commercial “Soap” Detergent Products
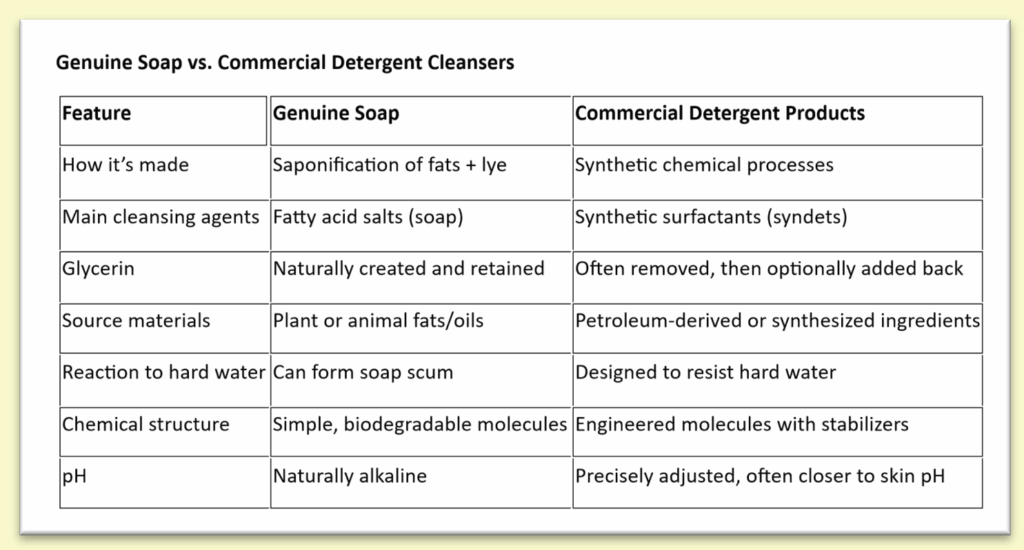
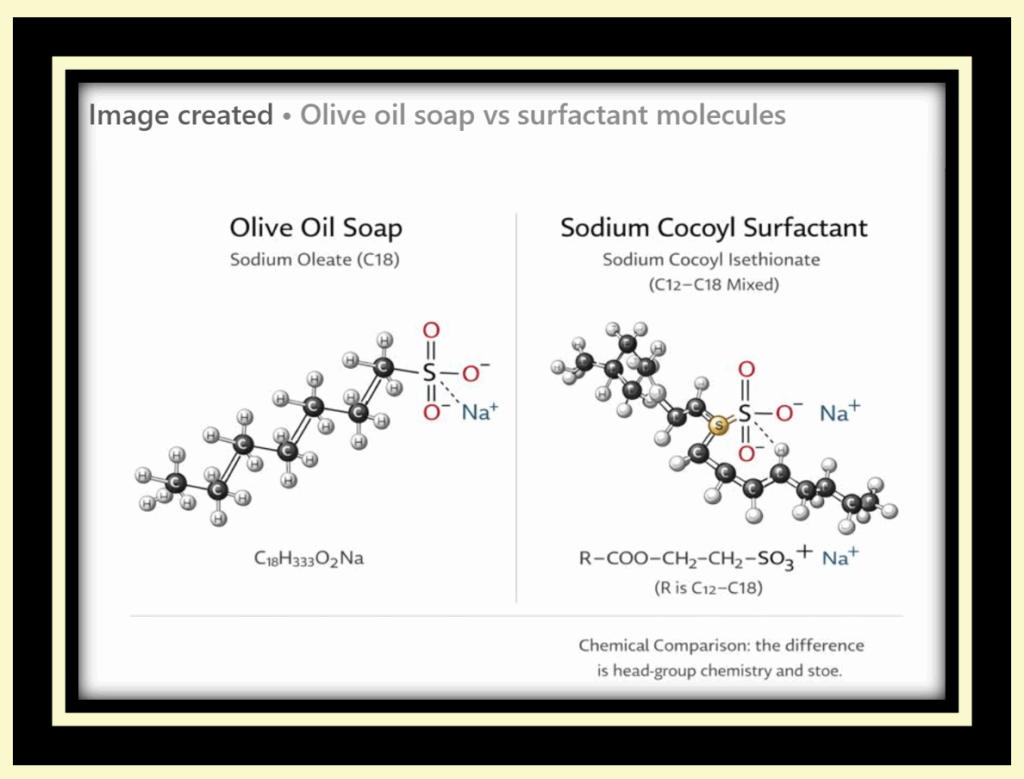
Chemically, genuine soap and most commercial cleansing products are fundamentally different substances. True soap is made through saponification, a chemical reaction in which triglycerides (fats or oils) react with sodium hydroxide (NaOH) or potassium hydroxide (KOH) to form soap molecules (fatty acid salts) and glycerol. These soap molecules are naturally biodegradable surfactants with a polar head and a nonpolar tail.
Most commercial “soap” products, however, are not soap at all from a chemical standpoint. They are formulated using synthetic detergents (syndets), which are produced through industrial chemical processes rather than saponification. These detergents are often petroleum-derived or synthesized fatty alcohol–based surfactants, combined with chelators, preservatives, thickeners, and pH adjusters. Because true soap reacts with minerals in hard water and has a naturally higher pH, detergent-based cleansers are often chosen for consistency, shelf stability, and precise pH control rather than chemical simplicity.
The Science of Soap
Soap is a rinse-off cleansing product. Its job is to remove dirt, oils, and soils from the skin so they can be rinsed away with water. It is not designed to treat the skin, change skin function, or deliver nutrients. I believe understanding what soap actually does — and what it does not do — is an important part of choosing products honestly.
How Soap Is Made
Soap is made through a chemical reaction called saponification, where oils or fats react with lye and heat. During this reaction, the fats are converted into soap molecules and glycerin. Once saponification is complete, the oils used to calculate the lye amount no longer exist as oils — they have been chemically transformed into soap.
The soap molecules created by this reaction are what provide the cleansing action. They lift oils and soils from the skin and allow them to be rinsed away with water. Glycerin is also formed during this process and remains naturally present throughout the finished soap.
A chemical reaction in which fats or oils react with lye to form soapand glycerin.
Lye is sodium or potassium hydroxide, a necessary ingredient for making real soap. It is consumed during the reaction and does not remain as lye in finished soap
Handmade soap is produced in small batches, so natural variation can occur.
About Superfatting (Intentional and Controlled)
My soaps are intentionally formulated with a small superfat (5% or less), meaning the recipe contains slightly more oil than is required for the lye to react completely. This ensures that all of the lye is consumed during saponification and that the finished soap is not lye-heavy. Any oil that remains unreacted is present in very small amounts and is randomly dispersed throughout the soap matrix.
Superfatting does not mean that specific oils remain intact or preserved as functional skincare ingredients. During saponification, the vast majority of oils are converted into soap, with only a minimal excess remaining by design as a safety and balance measure. Because soap is a rinse-off product, this small amount of dispersed oil serves primarily to ensure proper chemical completion rather than to provide targeted cosmetic benefits.
A small excess of oil (typically 5% or less) used to ensure complete lye consumption and prevent lye-heavy soap.
Why Glycerin Is Present in Handmade Soap
Glycerin is naturally created during the soapmaking process as part of the saponification reaction. In handmade soap, this glycerin remains within the soap matrix. It is not added separately and is not a coating on the bar — it is simply part of the finished soap as produced by the chemistry of the reaction.
A naturally occurring byproduct of soapmaking that remains in handmade soap after saponification.
How Soap Actually Cleans
Soap molecules have two distinct parts:
- A water-attracting (polar) head
- A water-repelling (nonpolar) tail
When soap is used with water, the nonpolar tails attach to oils and soils on the skin, while the polar heads remain attracted to water. This allows the soap to surround and lift away dirt so it can be rinsed down the drain.
A substance that helps oil and water interact, allowing dirt and oils to be lifted and rinsed away.
Soap vs. Commercial “Soap” Products
From a chemistry standpoint, genuine soap and many commercial cleansing products are not the same thing.
True soap is made through saponification using fats or oils and lye. Many commercial products labeled as “soap” are actually made using synthetic detergents, which are produced through different chemical processes. These detergents are designed to clean well, work consistently in hard water, and remain stable on store shelves.
Both genuine soap and detergent-based cleansers are intended to clean — they are simply made differently and contain different types of cleansing molecules.
Lab-created cleansing agents commonly used in commercial cleansers, chemically distinct from true soap.
About pH and Soap
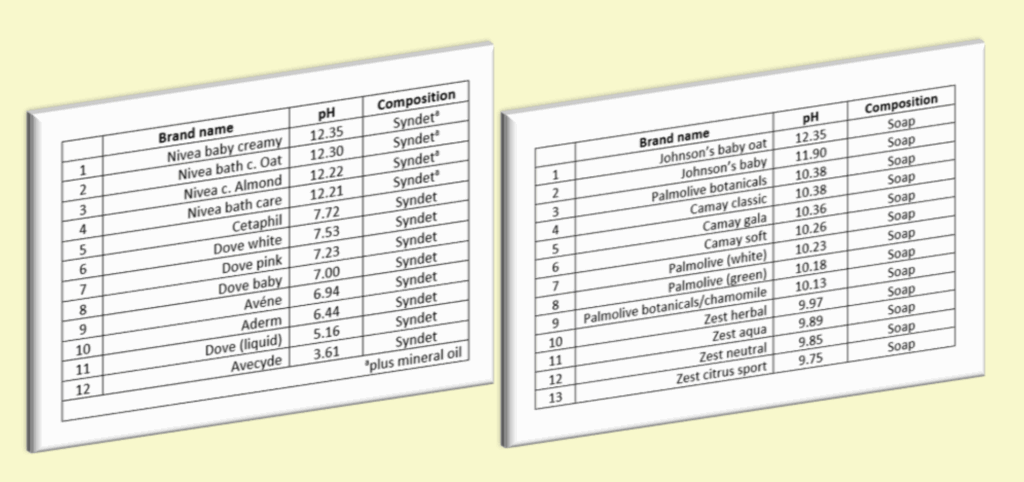
Soap is naturally alkaline due to its chemical structure. This is normal and expected for true soap. Detergent-based cleansers can be formulated at a wider range of pH levels, which is one reason they are often used in mass-produced products.
A measure of acidity or alkalinity. True soap is naturally alkaline.
Soap, Skin pH, and a Common Misconception
Because soap is a rinse-off product and the skin is a living, self-regulating organ, its naturally acidic surface is only temporarily altered during cleansing — much like it is by water alone. To explore why claims that real soap permanently damages the skin’s acidic mantle are misleading, and how the skin restores its balance, click here to learn more.
Why I Avoid Skin or Medical Claims
Soap is a rinse-off product. While it cleans effectively, it is not designed to treat, heal, or alter the skin. I focus on making well-balanced, thoughtfully formulated soap and providing accurate information so customers can make informed choices without exaggerated claims or marketing language.
Soap sold with cosmetic claims–moisturizing, etc–must be marketed as a “cosmetic” and fall under FDA guidelines.
My Philosophy
I believe transparency matters. That means explaining how soap works honestly, acknowledging what it does well, and being clear about its limitations. Our goal is not to convince anyone — it’s to inform, so you can decide what works best for you.
How Detergent-Based Cleansers Are Made
Detergent-based cleansers are not made the same way as soap. Instead of being created from fats and lye through a chemical reaction, detergents are manufactured using industrial chemical processes that build cleansing molecules from simpler raw materials.
Rather than transforming oils into soap, detergent molecules are designed and assembled to have the same basic cleaning ability: one part that grabs oil and dirt, and another part that mixes easily with water.
What Detergent-Based Cleansers Are Made From
In simple terms, detergents are made from:
Basic chemical building blocks, often derived from petroleum or from processed plant materials
Fatty alcohols (these are not drinking alcohols), which provide the oil-loving part of the molecule
Sulfur- or oxygen-based compounds, which create the water-loving part of the molecule
These components are chemically reacted to form synthetic surfactants — the actual cleansing agents in detergent-based products.
How They Are Manufactured
Raw materials are processed
Chemical feedstocks (often petroleum-based or plant-derived) are refined into simpler chemical components.Cleansing molecules are built
These components are chemically combined to create surfactants — molecules designed to attract both oil and water.Formulas are adjusted
Manufacturers then fine-tune the product by adjusting things like:thickness
fragrance
color
pH
shelf stability
The finished cleanser is blended
The surfactants are mixed with water and other ingredients to produce liquid body washes, shampoos, or syndet bars.
How Detergents Clean
Like soap, detergent molecules have:
A water-loving end
An oil-loving end
This allows them to surround dirt and oils and rinse them away with water. The cleaning principle is similar — the chemistry used to create the molecule is different.
The Key Difference from Soap (Simply Put)
Soap is made by transforming oils with lye
Detergents are made by assembling synthetic cleansing molecules from chemical components
Both clean effectively. They are simply made in different ways, from different starting materials, using different chemistry.
In Summary: Soap and Detergents
Soap and detergent-based cleansers both clean effectively, but they are made using different chemistry: soap is created by transforming fats through a simple chemical reaction, while detergents are engineered surfactants built through industrial processes.
In large-scale manufacturing, these differences also influence how byproducts are handled—particularly glycerin, which is naturally formed during soapmaking but is often separated out of commercial soap and combo bars for processing or use elsewhere.
If you arrived here from one of my soap pages and would like to read the full story of how synthetic detergent cleansers evolved during the 20th century, click the button to begin at the start of this series.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.