Why Glycerin Is Separated in Large-Scale Soap and Combo Bar Manufacturing
Introduction: Why Glycerin Matters in Soapmaking
Glycerin is an inevitable result of true soapmaking. Whenever fats or oils are reacted with an alkali through saponification, glycerin is formed alongside soap. In small-scale or traditional soapmaking, this glycerin typically remains in the finished bar. In large-scale industrial manufacturing, however, glycerin is often separated from the soap mass. This separation is not accidental, nor is it unique to modern products—it is a deliberate chemical and economic decision rooted in manufacturing efficiency.
Understanding how and why glycerin is removed helps clarify an important distinction between traditional soap, combo bars, and industrial cleansing products.
What Glycerin Is and How It Forms
Glycerin (glycerol) is a small, water-soluble molecule released when triglycerides are broken apart during saponification. Each triglyceride molecule yields:
soap molecules (fatty acid salts), and
free glycerin as a byproduct
This means glycerin is not an additive in true soap—it is created automatically as part of the chemistry. Once formed, glycerin strongly associates with water and remains dissolved unless intentionally separated.
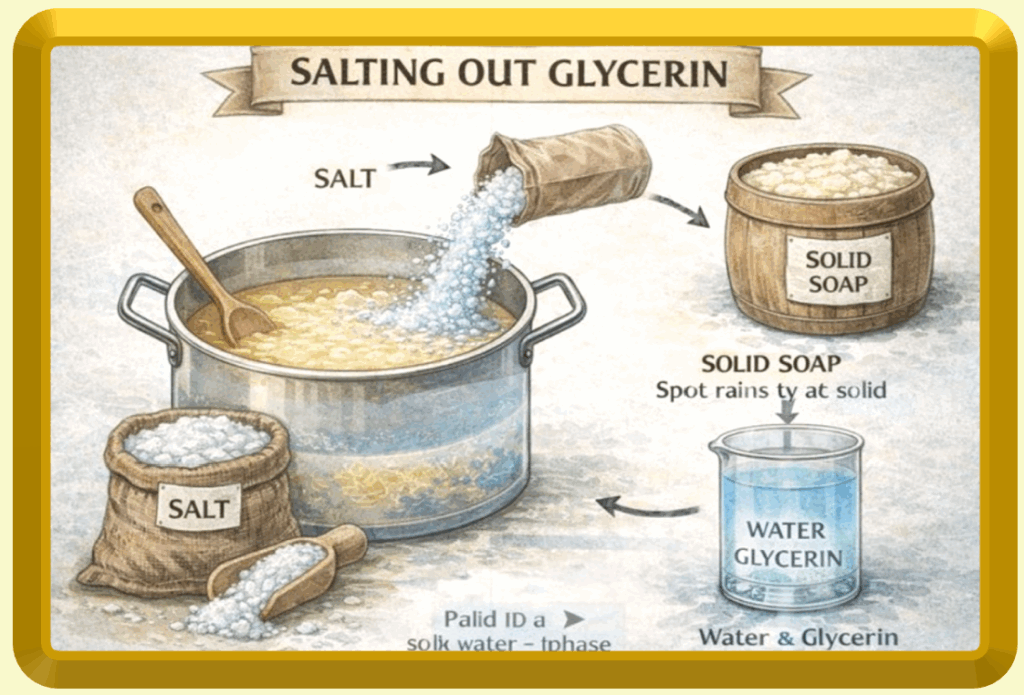
The Salting-Out Process: How Glycerin Is Removed
When large quantities of salt (typically sodium chloride) are added to freshly made soap:
soap becomes insoluble and precipitates out as a solid mass
glycerin remains dissolved in the salty water phase
This works because soap molecules are pushed out of solution by the salt, while glycerin stays behind due to its strong affinity for water.
What Happens Next
After salting out:
the solid soap is removed
the remaining liquid contains glycerin, water, salt, and trace impurities
This liquid is then further processed to:
recover glycerin
purify it to cosmetic, pharmaceutical, or technical grade
The result is a separate glycerin stream that can be sold or reused independently of the soap
Why Large Manufacturers Separate Glycerin
In large-scale soap and combo bar manufacturing, glycerin is often separated out for practical and economic reasons. Removing glycerin allows manufacturers to better control bar hardness, drying time, and shelf stability, which is especially important when producing consistent products across massive batches. Because glycerin attracts moisture, retaining it can complicate storage, handling, and long-term product uniformity.
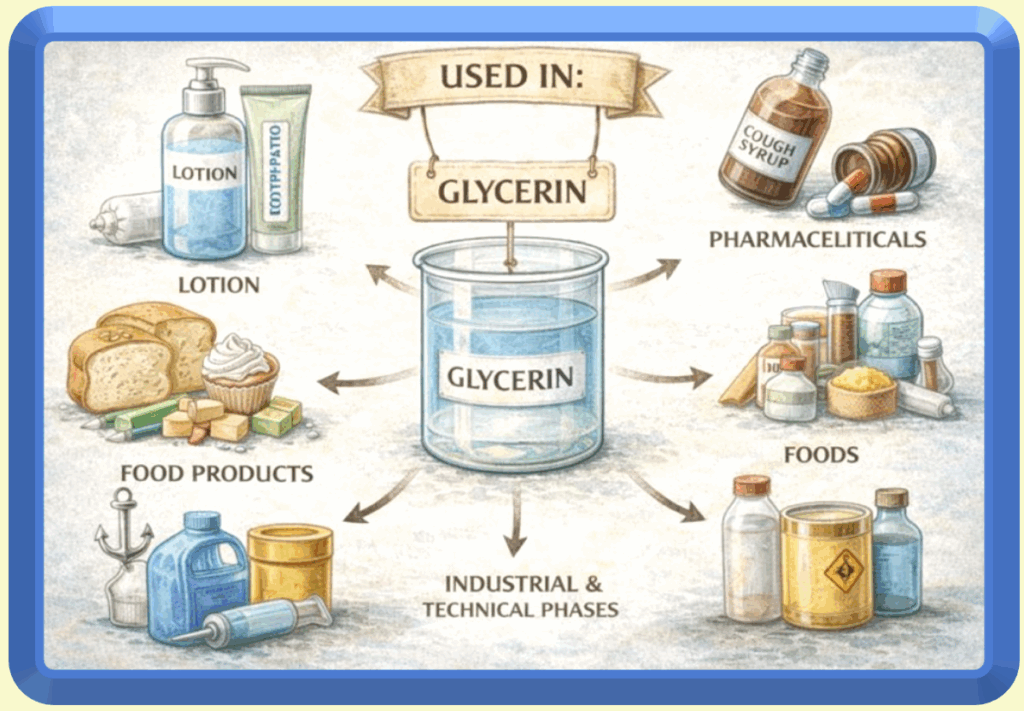
Beyond manufacturing considerations, glycerin itself is a valuable byproduct. Once separated and purified, it can be sold or redirected into other products such as lotions, toothpaste, pharmaceuticals, food applications, and industrial formulations. In many cases, glycerin has greater value when used separately than when left in a rinse-off cleansing bar. These decisions reflect scale, efficiency, and material optimization rather than product quality.
Glycerin in Combo Bars and Synthetic Detergent Systems
Combo bars—those containing both soap and synthetic detergents—do not chemically require glycerin to function. Synthetic surfactants cleanse effectively without it, and glycerin offers limited functional benefit in a detergent-forward system.
For this reason:
glycerin is often removed early
rarely added back
and typically reserved for other formulations
When glycerin is present in such bars, it is usually added intentionally and labeled accordingly, rather than being naturally retained.
Traditional Soap vs. Industrial Soap: A Structural Difference
In small-batch or handmade soapmaking:
glycerin is not removed
no salting-out step is used
the soap retains all naturally formed glycerin
This difference is structural, not moral or marketing-based. It reflects scale, process, and purpose rather than quality claims.
Why combo bars especially don’t get glycerin back
Combo bars are formulated with a strong emphasis on detergent performance and manufacturing efficiency. Because synthetic detergents do not require glycerin to function effectively, retaining glycerin offers little functional advantage in a detergent-forward system. Instead, combo bars are designed to perform reliably in hard water, maintain firmness, and remain stable over long shelf lives.
For these reasons, glycerin is often removed early in the production of combo bars and is rarely added back unless it serves a specific formulation purpose. From a formulation standpoint, glycerin is optional; from a business standpoint, it is often better utilized elsewhere. This is why most combo bars do not retain naturally formed glycerin and why claims emphasizing glycerin retention reflect marketing choices rather than standard industry practice.
Most large manufacturers follow the money and efficiency.
That means:
glycerin is removed
sold or repurposed
or used in higher-margin leave-on products
—not quietly returned to a combo bar where its benefit is minimal and its opportunity cost is high.
This is also why claims like “our bar retains its natural glycerin” are marketing choices, not industry norms — and why handmade soap is structurally different from combo bars in more ways than just ingredients.
Conclusion: Chemistry, Not Marketing
The separation of glycerin from soap and combo bars is a long-established industrial practice driven by chemistry, scale, and economics. Through salting out and downstream purification, manufacturers recover glycerin as a versatile and valuable material, often redirecting it into other products or markets.
Understanding this process helps clarify why glycerin content varies across cleansing products—and why its presence or absence reflects manufacturing choices rather than a universal standard. In the end, glycerin separation is not about what soap should be, but about how different systems are designed to operate.
If you arrived here from one of my soap pages and would like to read the full story of how synthetic detergent cleansers evolved during the 20th century, click the button to begin at the start of this series.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.