Coal to Clean
Did Germany Make Detergents from Coal?
How coal fit into detergent production
Coal was processed into coal tar, synthesis gas, and aromatic intermediates, which were then chemically transformed into:
Long-chain hydrocarbons
Aromatic compounds (benzene-based structures)
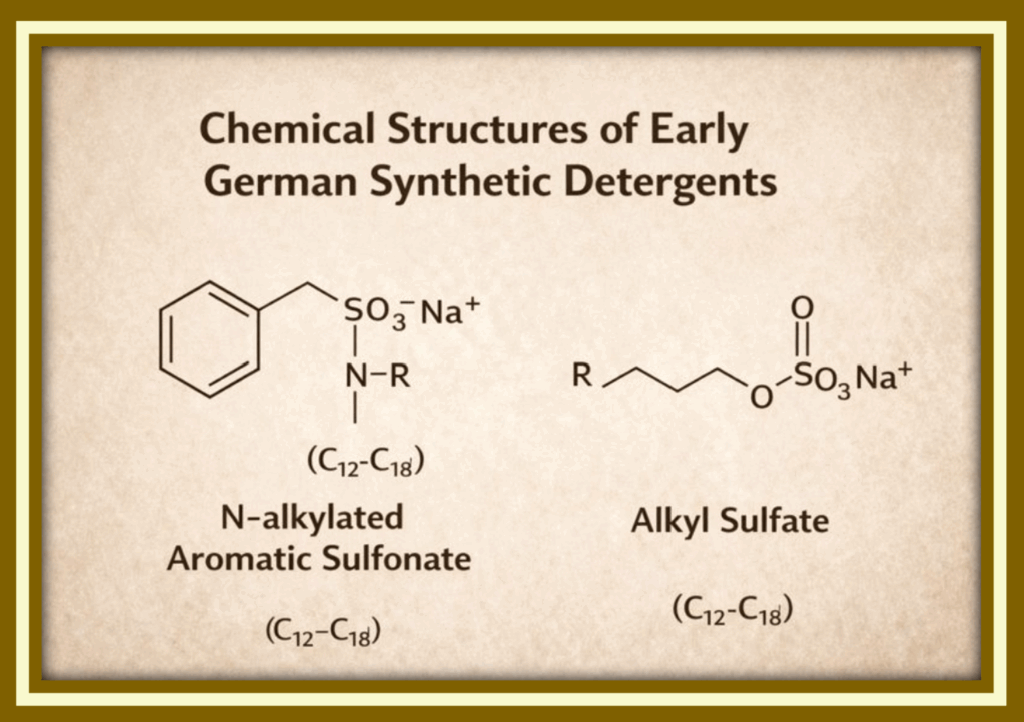
Sulfonated and sulfated surfactants
These compounds became the hydrophobic “tails” and charged “heads” that define synthetic detergents.
In other words:
Coal replaced fats and oils as the carbon source.”
Why coal mattered so much (especially in Germany)
During WWI and WWII, Germany faced:
Severe shortages of animal fats
Loss of vegetable and tropical oils
Blockades that cut off traditional soap materials
Coal, however, was:
Abundant domestically
Already central to German chemical industry
Chemically versatile
This made coal the logical foundation for fat-free detergents.”
Were coal-based detergents actually successful?
Yes — and decisively so.
Coal-derived chemistry enabled detergents that:
Could be produced without triglycerides
Scaled to industrial and military use
This success is why:
Synthetic detergents did not disappear after the wars
The technology spread internationally
Later petroleum-based detergents evolved directly from this chemistry
Coal-derived chemistry enabled detergents that could be produced without triglycerides and scaled efficiently for industrial and military use, making them practical substitutes for soap under wartime conditions.
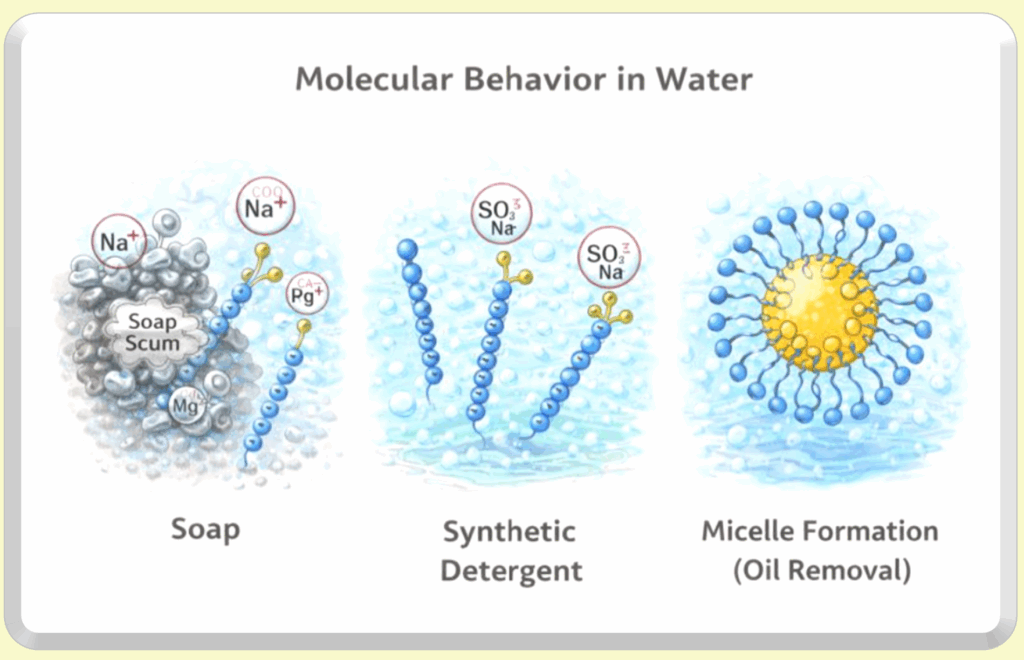
Visual Comparison: Soap vs Synthetic Detergents
Soap molecules have a water-loving (hydrophilic) head and an oil-loving (lipophilic) tail. In soft water, soap can form micelles that surround and lift away oils. In hard water, however, soap reacts with minerals like calcium and magnesium, forming insoluble salts (soap scum) that reduce cleansing efficiency and leave residue behind.
Synthetic detergents also have a hydrophilic head and a hydrophobic tail, but their head groups (such as sulfonates or sulfates) remain stable in hard water. This allows detergent molecules to consistently form micelles that trap oils and soils, keeping them suspended in water so they can be rinsed away without forming residue.
Micelle Formation (Oil Removal)
When enough soap or detergent molecules are present, they self-assemble into micelles. The oil-loving tails cluster inward around grease or dirt, while the water-loving heads face outward toward the surrounding water. This structure allows oily soils to be lifted from surfaces and carried away during rinsing.
Why Petroleum Replaced Coal (Even Though the Chemistry Stayed the Same)
Early synthetic detergents were developed using coal-derived chemical intermediates because coal was abundant and accessible, especially in Germany during wartime. The underlying chemistry of detergents—building long hydrocarbon chains and attaching sulfate or sulfonate head groups—was already established. What changed over time was not the chemistry itself, but the source of those carbon chains.
As petroleum refining expanded, it provided a more consistent, flexible, and economical source of hydrocarbons. Petroleum could be fractionated into predictable chain lengths ideal for surfactant production, reducing processing complexity and cost. In contrast, coal-derived chemistry required multiple conversion steps to obtain suitable intermediates. Once petroleum became widely available, manufacturers adopted it as a feedstock while continuing to use the same detergent chemistry, simply applied to a different raw material base.
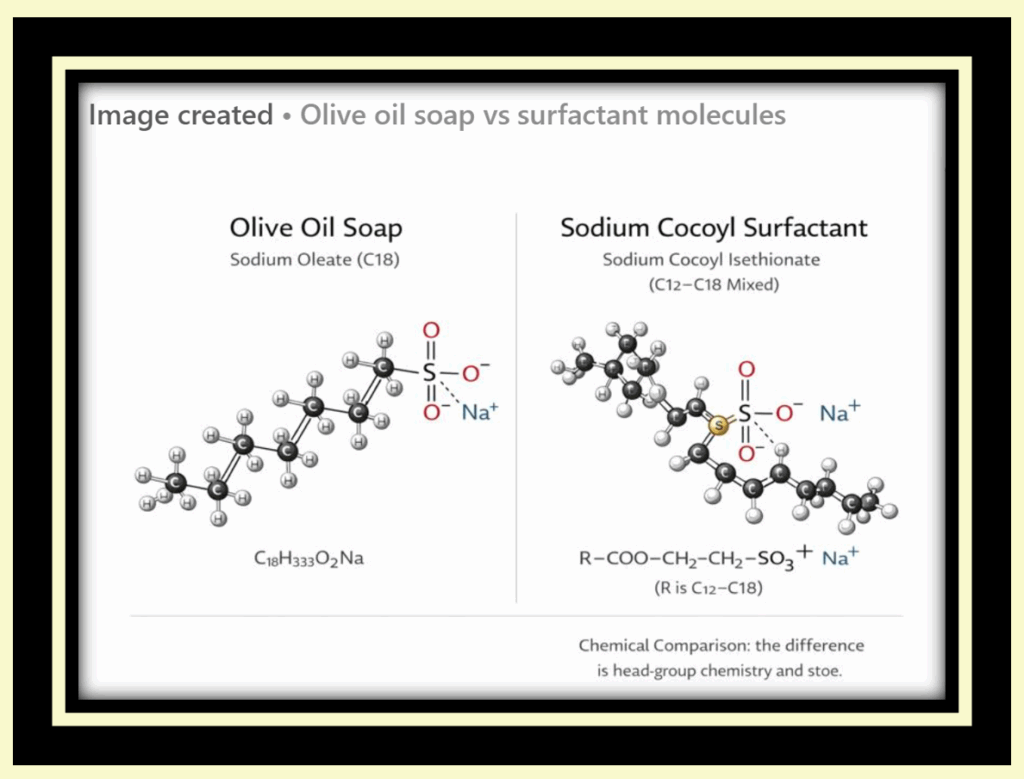
Soap vs. Synthetic Detergents: Chemistry and Behavior in Water
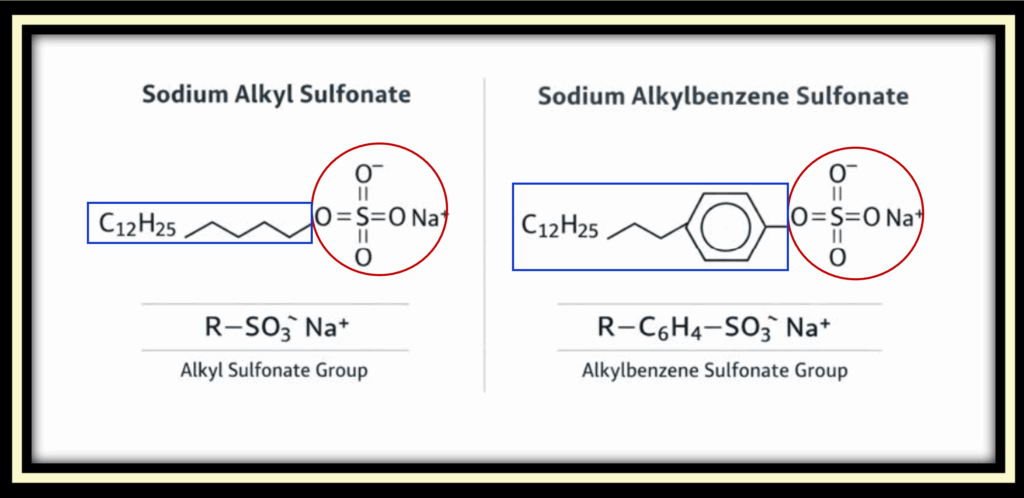
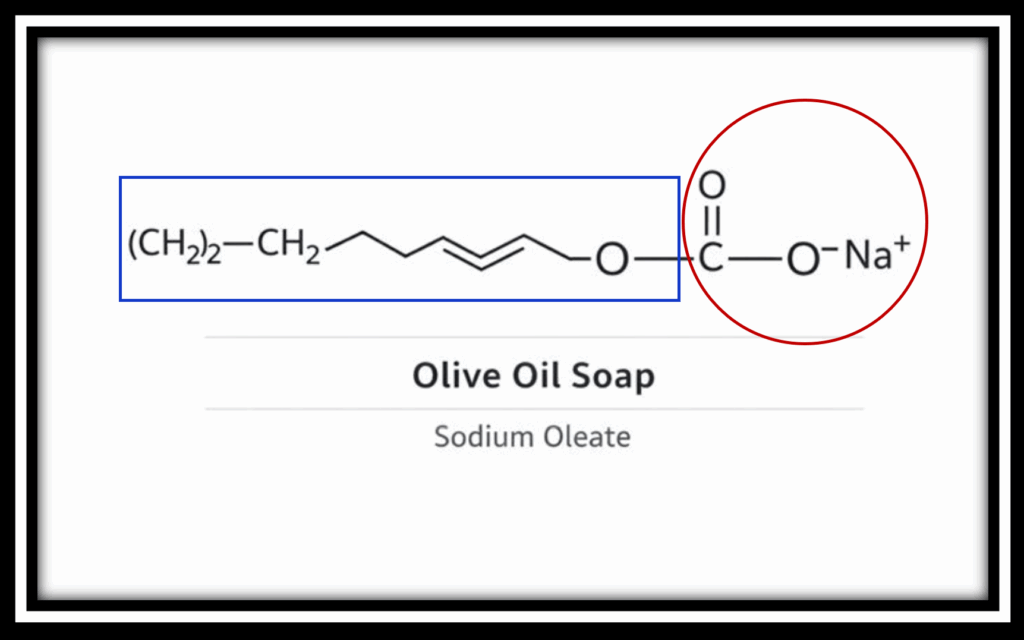
Traditional olive oil soap is composed of fatty acid salts, such as sodium oleate, formed from natural fats combined with alkali. Its chemical structure includes a carboxylate head group (–COO⁻ Na⁺ circled in red) attached to a long fatty acid tail (in blue rectangle). While this structure allows soap to lift and remove oils in soft water, it reacts readily with dissolved minerals such as calcium and magnesium found in hard or mineral-rich water. In these conditions, soap forms insoluble salts which reduces lather, diminishes cleaning efficiency, and leaves residue behind.
Synthetic detergents differ fundamentally in their chemical makeup. Instead of carboxylate head groups, they use sulfate or sulfonate head groups (such as –SO₃⁻ Na⁺ circled in red), attached to hydrocarbon tails derived from coal or petroleum sources (in the blue rectangle). These head groups remain stable in hard water and saline environments, allowing detergent molecules to form micelles consistently without precipitating out of solution. As a result, detergents continue to lather and clean effectively in conditions where soap chemistry fails.
The images below highlight these structural differences, showing the distinct head groups and hydrocarbon tails that determine how soap and synthetic detergents behave in water.
Why This Difference Mattered in Germany
The chemical differences between soap and synthetic detergents become clearest when viewed at the molecular level.
As mentioned earlier, Germany faced severe shortages of essential raw materials for soap production during wartime. At the same time, much of the available water was mineral-rich, further limiting soap’s practical use. Synthetic detergents offered a solution on both fronts: they did not rely on fats or oils, and they performed reliably under conditions where traditional soap was no longer practical.
The shift from soap to synthetic detergents in Germany was therefore driven not by preference or gentleness, but by chemistry and necessity. Detergents succeeded where soap chemistry could not, fundamentally changing how cleaning agents were produced and used under wartime conditions—and setting the stage for their continued use long after the shortages ended.
Conclusion: From Coal to Clean
The development of synthetic detergents began with coal not because it was ideal, but because it was available. Coal-derived chemistry allowed German chemists to replace fats and oils with synthetic carbon chains and create detergents that functioned in hard and saline water where traditional soap could not. Although effective, coal-based detergent production required multiple processing steps to obtain suitable intermediates, making it complex and resource-intensive.
As petroleum refining expanded, it offered a more direct and efficient source of the same hydrocarbon building blocks. This shift did not change the underlying chemistry of detergents—the same surfactant structures and cleaning mechanisms remained in use—but it simplified production and improved consistency. Over time, petrochemicals replaced coal as the preferred feedstock, not because detergents were reinvented, but because the path from raw material to finished cleaner became shorter and more economical. The transition from coal to petroleum reflects an evolution in industrial efficiency rather than a change in how detergents work.
If you arrived here from one of my soap pages and would like to read the full story of how synthetic detergent cleansers evolved during the 20th century, click the button to begin at the start of this series.
Educational & Informational Disclaimer
The information provided on this website is for educational and informational purposes only. It is intended to share general knowledge about traditional soapmaking, ingredient behavior, historical context, and manufacturing processes.
Nothing on this site is intended to diagnose, treat, cure, or prevent any disease or medical condition. The products offered are classified as soap under FDA guidelines and are not intended to affect the structure or function of the skin or body. Individual experiences and preferences may vary.
Information presented here should not be considered a substitute for professional medical advice, diagnosis, or treatment. If you have a medical condition, skin concern, or sensitivity, please consult a qualified healthcare professional before using any product.
This website may reference or link to third-party scientific, technical, or educational resources for general learning purposes. These references are provided to support transparency and encourage independent research, not as endorsements or medical guidance.
Click here to read the full disclaimer.
Contact me:
Text:
(479) 651-2760
I typically respond to texts almost immediately unless I’m in the middle of making soap.
As soon as I reach a stopping point, I’ll be sure to check and reply.
I check my email daily and will get back to you as soon as possible.